
Again, the mass of a mole substance is called the molar mass, and to find the molar mass of a substance you just need a periodic table and the chemical formula. Find out how to use the table for stoichiometry problems and conversion of units. That’s the total molar mass of the compound CO2. Learn about the elements, symbols, atomic numbers, and relative masses on the periodic table. Then finally you add all products, so 12 plus 32 is 44. The calculated value makes sense because it is almost four times times the mass for 1 mole of aluminum. You would multiply it by its subscript of 2, which is 32. Prepare a concept map and use the proper conversion factor. If the mass of a substance is known, the number of moles in the substance can be calculated. Then you’d do the same thing with oxygen. The molar mass of an element is found on the periodic table, and it is the elements atomic weight in grams/mole (g/mol). Then the way you would find that is first you would find the atomic mass of carbon, which is 12, and then you’d multiply it by the subscript, which is just an understood 1 right there, so the product there would be 12. If you were trying to find the molar mass of the compound carbon dioxide, which looks like this-CO2-and the molar mass is 44. Then, to find the molar mass of the compound, you’re going to add all those products together. By multiplying the atomic mass by the subscript you’re getting the product, you’re getting the molar mass product for that atom. That way, you’re finding the total molar mass for all of that atom because you’re finding the atomic mass, then you’re multiplying it by however many times it appears in the compound. After you’ve found the atomic mass, multiply the atomic mass by the subscript. You’re going to start with one atom at a time. I’m going to show you a 3-step process for finding the molar mass of a compound.įirst, find the atomic mass of an atom. You kind of follow the same protocol for finding the molar mass of an element, but then you combine all those molar masses together to find the molar mass of the compound. 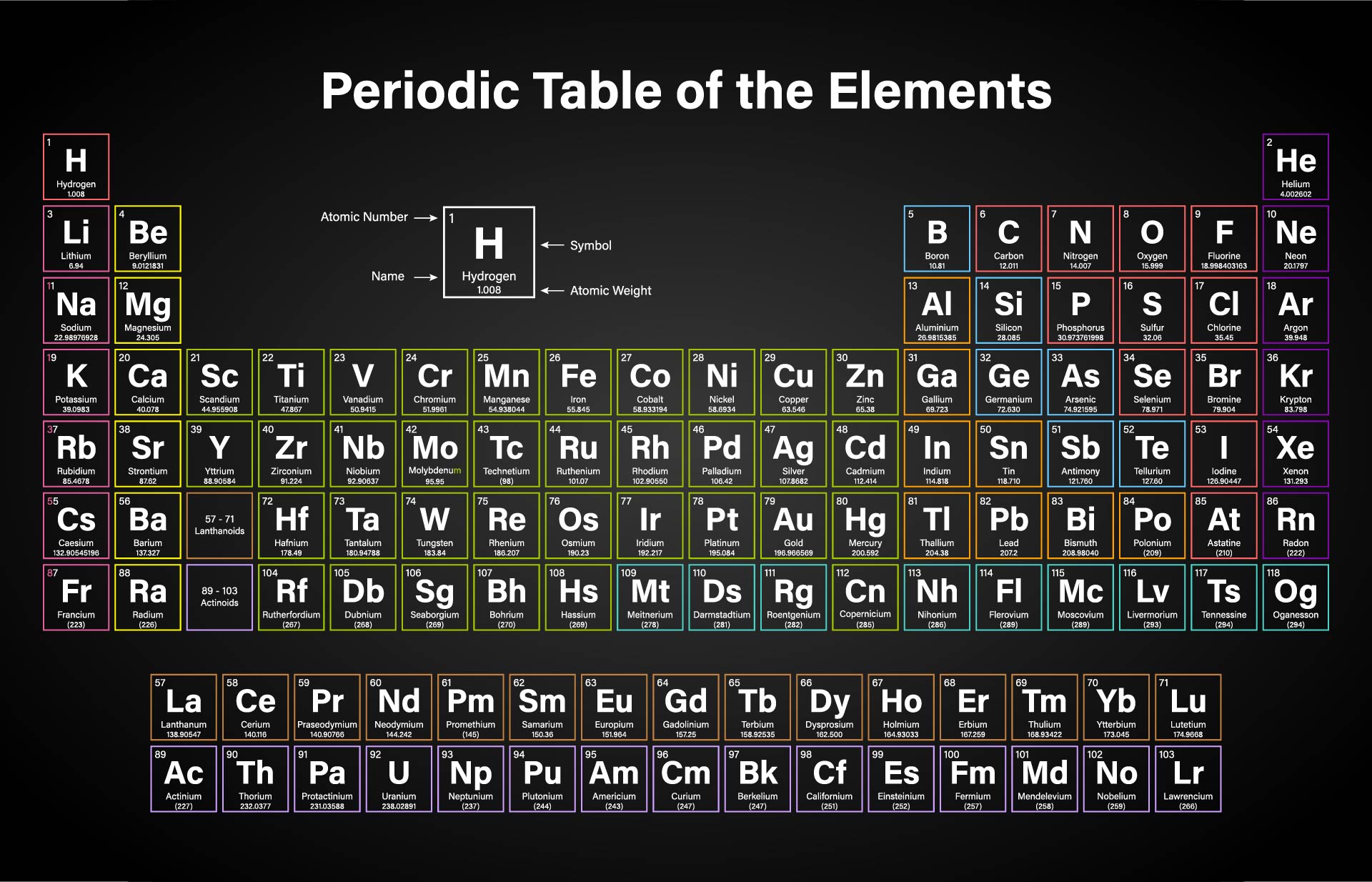
Now the molar mass of a compound can be calculated by summing the molar masses of each atom and the chemical formula. That’s how you find the molar mass of an element. It is a constant property of each substance for example, the. Molar mass is the mass of 1 mole of the solute. We can link the number of entities X in a specified sample N(X). Now we’ll write it with a different unit because it’s also our molar mass, and that will be grams over moles: \(\frac\). Moles allow you to directly read weight from the periodic table (e.g., 1 mole of N is 28 g or 1 mole of NaCl is 58.5 g). If you’re looking at the element of copper you would look in the periodic table and find that it has an average atomic mass of 63.55 AMU, so that’s the molar mass. If you’re trying to find the molar mass of an element, then you’ll take the average atomic mass listed in the periodic table. To calculate the molar mass of a substance you need a periodic table and the chemical formula. You can search for any element by its symbol, name, or atomic number, and see its molar mass, weight, energy levels, electronegativity, and more. Thus, the molar mass of bilirubin can be expressed as 584.73 g/mol, which is read as “five hundred eighty four point seventy three grams per mole.The mass of a mole of substance is called the molar mass. Ptable is a comprehensive online resource for the periodic table of the chemical elements, with detailed information on their properties, isotopes, isotopes, and isotopes. The division sign (/) implies “per,” and “1” is implied in the denominator. This is a periodic table with atomic mass, element name, element symbol, and atomic number. For example, the molar mass of Ba(OH) 2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: The molar mass of Ba(OH)2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: 1 Ba molar mass:īecause molar mass is defined as the mass for 1 mol of a substance, we can refer to molar mass as grams per mole (g/mol). In formulas with polyatomic ions in parentheses, the subscript outside the parentheses is applied to every atom inside the parentheses. adenosine (C 10H 13N 5O 4), a component of cell nuclei crucial for cell divisionīe careful when counting atoms.barium sulfate (BaSO 4), used to take X rays of the gastrointestional tract.What is the mass of 1 mol of each substance? \): Moles to Mass Conversion with Compounds
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
